This is an old revision of this page, as edited by PixelBot (talk | contribs) at 01:57, 27 October 2011 (r2.6.4) (Robot: Removing sl:Barijev karbonat). The present address (URL) is a permanent link to this revision, which may differ significantly from the current revision.
Revision as of 01:57, 27 October 2011 by PixelBot (talk | contribs) (r2.6.4) (Robot: Removing sl:Barijev karbonat)(diff) ← Previous revision | Latest revision (diff) | Newer revision → (diff)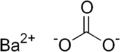 | |
 | |
| Names | |
|---|---|
| Other names Witherite | |
| Identifiers | |
| CAS Number | |
| 3D model (JSmol) | |
| ChemSpider | |
| ECHA InfoCard | 100.007.426 |
| EC Number |
|
| RTECS number |
|
| UNII | |
| UN number | 1564 |
| CompTox Dashboard (EPA) | |
InChI
| |
SMILES
| |
| Properties | |
| Chemical formula | BaCO3 |
| Molar mass | 197.34 g/mol |
| Appearance | white crystals |
| Density | 4.286 g/cm |
| Melting point | 811 °C |
| Boiling point | 1360 °C (decomp) |
| Solubility in water | 0.0024 g/100 mL (20 °C) |
| Solubility | soluble in HCl, HNO3, ethanol insoluble in H2SO4 |
| Refractive index (nD) | 1.60 |
| Hazards | |
| NFPA 704 (fire diamond) |
 |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
| LD50 (median dose) | 418 mg/kg, oral (rat) |
| Related compounds | |
| Other cations | Magnesium carbonate Calcium carbonate Strontium carbonate |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Barium carbonate (BaCO3), also known as witherite, is a chemical compound used in rat poison, bricks, ceramic glazes and cement.
Witherite crystallizes in the orthorhombic system. The crystals are invariably twinned together in groups of three, giving rise to pseudo-hexagonal forms somewhat resembling bipyramidal crystals of quartz, the faces are usually rough and striated horizontally.

The mineral is named after William Withering, who in 1784 recognized it to be chemically distinct from barytes. It occurs in veins of lead ore at Hexham in Northumberland, Alston in Cumbria, Anglezarke, near Chorley in Lancashire and a few other localities. Witherite is readily altered to barium sulfate by the action of water containing calcium sulfate in solution and crystals are therefore frequently encrusted with barytes. It is the chief source of barium salts and is mined in considerable amounts in Northumberland. It is used for the preparation of rat poison, in the manufacture of glass and porcelain, and formerly for refining sugar. It is also used for controlling the chromate to sulfate ratio in chromium electroplating baths.
Preparation
Barium carbonate is made commercially from barium sulfide either by treatment with sodium carbonate at 60 to 70 °C (soda ash method) or by passing carbon dioxide at 40 to 90 °C.
In the soda ash process, solid or dissolved sodium carbonate is added to barium sulfide solution, and the barium carbonate precipitate is filtered, washed and dried.
Reactions
Barium carbonate reacts with acids such as hydrochloric acid to soluble barium salts, such as barium chloride:
- BaCO
3 (s) + 2 HCl (aq) → BaCl
2 (aq) + CO
2 (g) + H
2O (l)
However, the reaction with sulfuric acid is poor, because barium sulfate is highly insoluble.
Uses
Barium carbonate is widely used in the ceramics industry as an ingredient in glazes. It acts as a flux, a matting and crystallizing agent and combines with certain coloring oxides to produce unique colors not easily attainable by other means. Its use is somewhat controversial since some claim that it can leach from glazes into food and drink. To provide a safe means of use, BaO is often used in fritted form.
In the brick, tile, earthenware and pottery industries barium carbonate is added to clays to precipitate soluble salts (calcium sulfate and magnesium sulfate) that cause efflorescence.
References
- Whitelaw, G.P. (2003-10-25). "Standard Chrome Bath Control". finishing.com. Retrieved 2006-11-29.
{{cite web}}: Cite has empty unknown parameter:|coauthors=(help) - Pradyot Patnaik. Handbook of Inorganic Chemicals. McGraw-Hill, 2002, ISBN 0070494398
 This article incorporates text from a publication now in the public domain: Chisholm, Hugh, ed. (1911). Encyclopædia Britannica (11th ed.). Cambridge University Press.
This article incorporates text from a publication now in the public domain: Chisholm, Hugh, ed. (1911). Encyclopædia Britannica (11th ed.). Cambridge University Press. {{cite encyclopedia}}: Missing or empty|title=(help)
| Pest control: Rodenticides | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Anticoagulants / Vitamin K antagonists |
| ||||||||
| Convulsants | |||||||||
| Calciferols | |||||||||
| Inorganic compounds | |||||||||
| Organochlorine | |||||||||
| Organophosphorus | |||||||||
| Carbamates | |||||||||
| Others | |||||||||